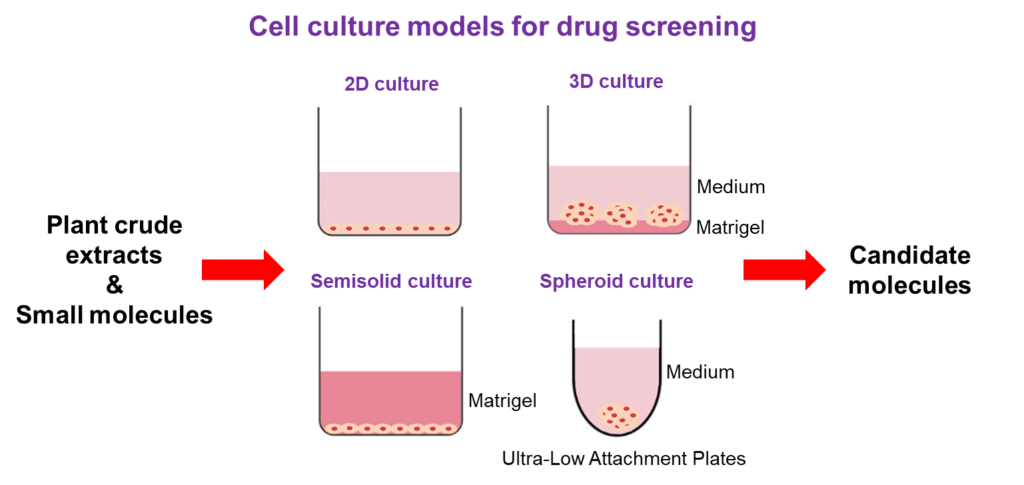
Tumor microenvironment
It is now widely accepted that the tumor microenvironment influences the fate and behavior of cancer cells and plays crucial roles in regulating tumor progression and therapeutic responses. The microenvironment of cells cultured in vitro on traditional plastic substrata does not account for cell interactions with the extracellular matrix (ECM) and is thus a less reliable approach to analyze cellular activity. However, most studies are performed with cancer cells grown in two-dimensions on plastic plates, leading to high failure rate of drug discovery and development. Culture conditions that recreate the appropriate microenvironment for cells should provide a better model for studying cellular activity and discovery of anticancer drugs.
We thus developed cell culture models that more closely resemble in vivo tumor biology and investigated its consequences on cellular activity and responses to anticancer compounds. The results of an in vitro 3D model showed that 3D cultured cells on Matrigel showed cellular dormancy (G0/G1 cell cycle arrest), decreased cell migration and invasion, and were resistant towards a variety of chemotherapeutic agents.Further investigation of the molecular mechanisms implicated in tumor dormancy and drug resistance led to the identification of a key mediator involved in the development of chemoresistance in 3D cultures which may offer the potential of developing effective targeted therapies for cancer.