Cancer biology: metastasis, blood vessel formation and drug resistance
Metastasis and drug resistance of cancer cells are major problems in cancer therapy. Metastasis is the spread of cancer cells in the body, from primary tumor site to distant organs, where the cancer cells remain dormant for months to years as micrometastases, and then regrow to become recurrent tumors. We have explored the effect of chemicals and drugs on metastasis of cancer cell lines. A common environmental contaminant, perfluorooctanoic acid (PFOA), enhanced the metastatic potential of FTC133 thyroid cancer cells, while sub-lethal doses of the drug, doxorubicin, increased metastatic potential of low-invasive HepG2 liver cancer cells.
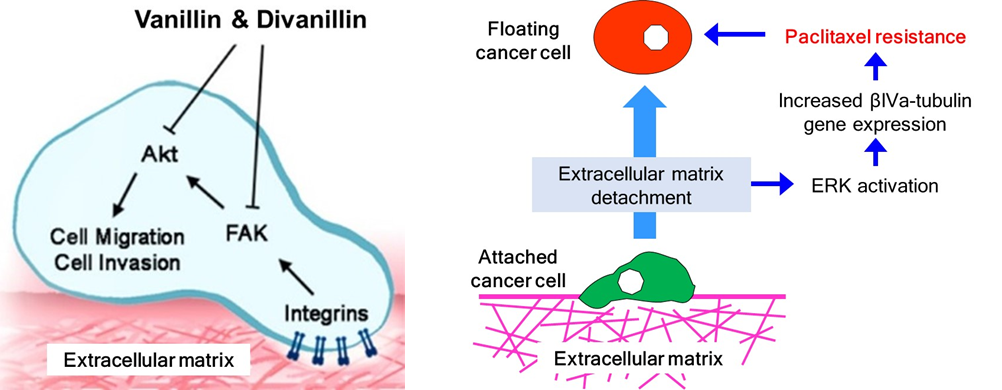
On the other hand, vanillin, the vanilla flavoring agent, was found to reduce the metastatic potential of 4T1 mouse breast cancer cells and to suppress metastasis in an animal model, orally given vanillin for a month. Vanillin and its structure-related compound, apocynin, decreased metastatic potential of A549 lung cancer cells by inhibiting the PI3K/Akt signaling pathway. Homodimers of vanillin and apocynin were more potent than their monomers in inhibiting invasion of HepG2 liver cancer cells, and protein focal-adhesion kinase (FAK) was identified as a target of the dimeric compounds. Chrysin, found in Thai propolis, also showed anti-metastatic potential by reducing tumor recurrence, through suppressing growth of breast cancer micrometastases after spreading to the lungs of mice.With chrysin, STAT3 inhibition was the mechanism for inhibiting hypoxia-induced VEGF expression and decreasing recurrence of metastatic breast cancer cells. Additionally, chrysin also sensitized cancer cells to TRAIL-induced apoptosis through inhibition of STAT3.
New blood vessel formation, both originating from pre-existing blood vessels (angiogenesis) and blood channel formation by cancer cell assembly (vasculogenic mimicry), require a cellular invasion process similar to cancer metastasis. Agents having anti-metastatic potential, Eclipta prostrata juice and vanillin, were found to inhibit angiogenesis in vivo by using a fertilized hen egg model. Vasculogenic mimicry (VM) capability of cancer cells was found to be associated with the intrinsic invasiveness/metastatic potential of the cancer cells. We also demonstrated that a pro-metastatic growth factor, hepatocyte growth factor (HGF), often found in the tumor microenvironment, could enhance the VM capability of non- invasive human liver cancer cells, HepG2, by induction of epithelial-mesenchymal transition (EMT) in the cells. Additionally, we found that curcumin showed anti-VM potential by inhibiting VM formation of SK-Hep-1 liver cancer cells, and also studied the underlying mechanism.
Understanding the mechanism of drug resistance is important for management of treatment and development of new anticancer drugs. We have three cellular models for development of drug resistance. The first is a drug resistant lung cancer cell line A549RT-eto, after long term culture of A549 cells in media containing an anticancer drug. The established cell line showed a cross-resistance against several chemotherapeutic drugs, and this multidrug resistance/MDR cell line has been used as a model for developing strategies for reversing drug resistance. The second model is culturing cancer cells under low oxygen condition (hypoxia) which occur in tumors when blood supply is insufficient, and adaptation of cancer cells to hypoxia often leads to increase in drug resistance and metastatic potential. Thus, hypoxic culture of HepG2 human liver cancer cells led to resistance to curcumin. The third model is floating cancer cells. Lung cancer cells can also develop drug resistance during metastasis. So, H460 human lung cancer cells were cultured under non-adherent conditions, to mimic the situation where cancer cells enter the blood or lymphatic circulatory systems during metastasis. The floating cancer cells rapidly developed resistance, specifically to a microtubule-targeting drug, paclitaxel, due to changes in the expression beta-tubulin isotype.